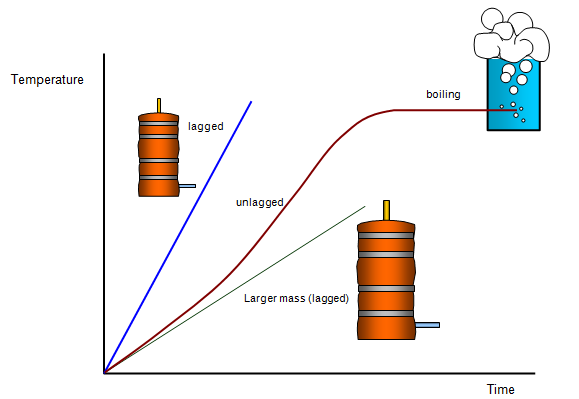

In all the problems that we have looked at so
far we have assumed that no heat is lost to the surroundings but this is not true. There will
always be some heat loss and the heat energy that we put into the water that we are heating
is always going to be a bit less than we think and so the rise inn temperature in a certain time
will also be less. Lagging the object will help and the following graph shows the effect of
doing this. The graph also shows how the graph levels off when the water boils.
(A
similar graph for heating a metal block with a heater but the leveling off would then show the
block melting and not boiling). (Find out more about melting and boiling by looking at other
sections.)