
In 1896 Henri Becquerel, who was Professor of Physics
at the University of Paris, discovered that uranium salts could fog a photographic plate even
if the plate was covered. He deduced that the salts were emitting some form of radiation that
could pass through the wrapping. He also found that this radiation could ionise a gas,
causing a charged electroscope to discharge.
In the same year Marie Curie and
Schmidt discovered that thorium also produced this radiation.

In 1898 Marie Curie extracted a much more active material from the
ore pitchblende, which is 80 per cent uranium oxide. She called the radiation radioactivity
and the active material radium. Radium is so active that its compounds stay a few degrees
above the temperature of their surroundings.
In 1900 the radioactive
gases thoron and radon were discovered by Rutherford and Dorn.
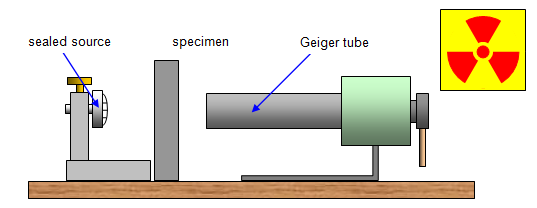
Properties of the radiation
It was later realised that this radiation:
(a) came from the nucleus of unstable atoms
(b) was spontaneous – once material containing these unstable nuclei was formed emission could take place immediately and without the addition of other types of energy
(c) was random – it was impossible to predict which nucleus would emit radiation
| Alpha radiation | Beta radiation (MeV) | Gamma radiation | |
| Penetration | small | large | very large |
| Range in air | few cm | tens of cm | many metres |
| Charge | +2 | +1 or -1 | 0 |
| Deflection by electric or magnetic fields |
some | large | none |
| Ionisation | 10000 | 1000 | 1 |
| Velocity | 0.05c | 0.9c | c |
| Mass | 8000 | 1 | '0' |
| Nature | helium nuclei | electrons | e.m radiation |
In a vacuum the range of either alpha, beta or gamma radiation is infinite, since
no ions are produced. The intensity of gamma radiation decreases with distance, following
the inverse square law since it is electromagnetic radiation.
Alpha particles have a
very short range in materials and their range is difficult to measure. The absorption of
gamma radiation can be studied quite easily, however, and it is found that the intensity of the
gamma radiation (I) varies with distance (t) through the material, according to the equation: