
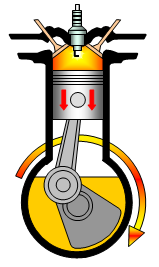
The study of thermodynamics resulted from the desire during the
industrial revolution to understand and improve the performance of heat engines such as the
steam engine and late the internal combustion engine.
This section contains many
references to heat and temperature so it is important to define these terms. Strictly
speaking:
When heat
energy is supplied to a gas two things may happen:
(a) the internal energy of the gas
may increase
(b) the gas may do external work
Considering this in another way, the
internal energy of a gas will increase if either:
(a) heat energy is added to it by heating it
or
(b) work is done on the gas by compressing it
This leads us to a
proposal know as the First Law of thermodynamics.
This means that
there is a finite amount of energy in the Universe and although this energy can be changed
from one form to another the total amount never changes – if we want to use energy in one
form then we have to 'pay for it' by converting it from energy in another form.
If we
consider the First Law in equation form as it applies to a gas then:
Increase in
internal energy (dU) = Heat energy supplied (dQ) + Work done on the gas (dW)
Note that dU represents both the change in the internal kinetic energy of the gas (an increase in molecular velocity) and the increase in the internal potential energy (due an increase in energy overcoming intermolecular forces due to separation of the molecules). The potential energy increase is zero for ideal gases and negligible for most real gases except at temperatures near liquefaction and/or at very high pressures.