
The following short list gives some important dates in the
discovery of the properties of the electron:
1869 Hittorf invented the Maltese cross
tube
1876 Goldstein first used the term cathode rays for the radiation that appeared to come
from the cathode in thermionic emission
1879 Crookes deflected the rays with a magnetic
field showing that they were charged particles and also determining their sign and direction of
flow
1897 Thomson showed that the value of the charge to mass ration (e/m) was always the
same he also assumed that the charge carried was the same as that in electrolysis and so
calculated the mass of the electron
1909 Millikan measured the electron charge
Discovered by Edison in 1880
As you may know, metals contain many free electrons moving at high speed and at random between the atoms of the material. If these free electrons are given enough energy they may actually escape from the metal surface.
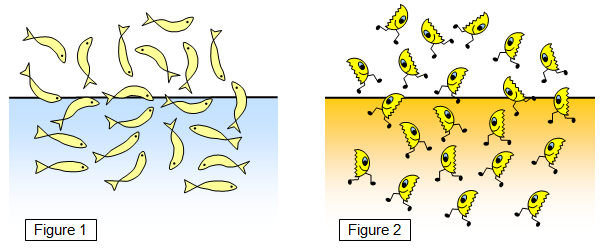
We
can compare the motion of the free electrons (Figure 2) with that of a pond full of fish (Figure 1).
The fish are crowded together in the pond and swim around vigorously - leaping in and out of the
water to give a dynamic equilibrium (as many fish are leaving the water at any time as are
returning to it).
If the energy of the fish is increased more of them will be leaping out and
falling back - the process will be come more violent. The faster they swim the more will leap out
and the further from the water will they get before they fall back in again. If some incentive is given
for them to leave the water for good - for example a worm - there will be a flow of fish away from
the pond!
One way to give the free electrons sufficient energy to allow them to escape is
to heat the metal. If you put a wire carrying a fairly large current (6A) close (within a couple of mm)
to the plate of a positively charged electroscope the emission of electrons from the wire will
discharge the electroscope.
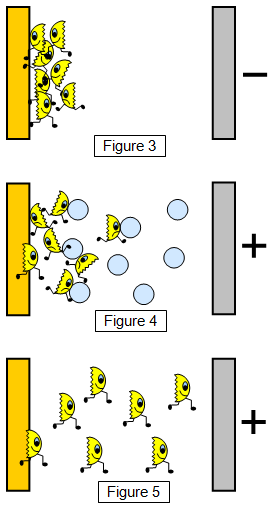
If we put a negative plate in front
of our hot metal any electrons that are emitted will be pushed back towards the
metal by the negative charge on the plate. None of them will get very far from
the metal. This happens because the electrons are also negatively
charged.
If we make the plate positive the electrons will
get attracted towards it, but if the space between the metal and the plate
contains air the electrons will not get far. They will collide with the
molecules of the air, and so very few will reach the positive plate. Hence the
reason for putting the hot wire close to the electroscope plate in the previous
experiment.
However if we put the metal and the plate in an
evacuated glass container the electrons will be pulled across. The hotter we
make the metal the more electrons will escape and travel to the positive plate.
Increasing the voltage on the plate will also increase the flow and make them
move faster.